The biologics CDMO secondary packaging market is witnessing strong expansion as the global demand for biologic drugs continues to rise. Contract Development and Manufacturing Organizations play a crucial role in supporting pharmaceutical companies by offering specialized packaging services that ensure product safety, regulatory compliance, and efficient distribution. Secondary packaging for biologics includes cartons, labels, inserts, and protective packaging solutions designed to maintain the integrity of sensitive biologic products.
Biologics such as vaccines, monoclonal antibodies, and cell and gene therapies require highly controlled packaging environments. As a result, CDMOs are investing in advanced secondary packaging technologies that offer traceability, temperature control compatibility, and regulatory adherence. The increasing outsourcing trend in the pharmaceutical industry is further driving the demand for biologics CDMO secondary packaging services.
Market Size
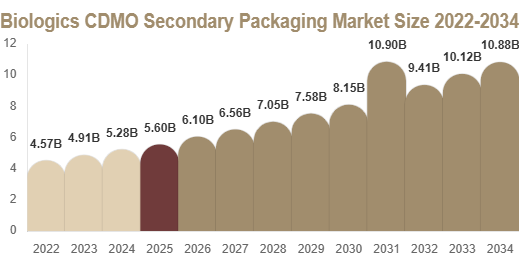
The global biologics CDMO secondary packaging market size was valued at USD 2.4 billion in 2025 and is projected to reach USD 4.6 billion by 2034.
The market is expected to expand at a CAGR of 7.5% during the forecast period from 2025 to 2034.
Get Your Sample Report Here: https://packagingmarketinsights.com/report/biologics-cdmo-secondary-packaging-market/request-sample
Market Overview
Biologics CDMO secondary packaging involves the packaging of biologic drugs after primary packaging, ensuring that products are ready for distribution while maintaining safety and compliance. These services include labeling, cartoning, serialization, and aggregation, which are essential for regulatory compliance and supply chain transparency.
The market is driven by the increasing complexity of biologic drugs, which require specialized handling and packaging solutions. CDMOs offer flexible and scalable packaging services, enabling pharmaceutical companies to focus on core competencies such as drug development and commercialization. Additionally, advancements in digital tracking and serialization technologies are enhancing the efficiency and reliability of secondary packaging processes.
Buy Now Full Report: https://packagingmarketinsights.com/report/biologics-cdmo-secondary-packaging-market
Market Drivers
Rising demand for biologic drugs
The increasing prevalence of chronic diseases and the growing adoption of biologic therapies are driving demand for specialized packaging solutions. Biologics require precise handling and packaging to maintain their efficacy, boosting the demand for CDMO services.
Growing trend of outsourcing packaging services
Pharmaceutical companies are increasingly outsourcing packaging operations to CDMOs to reduce costs and improve efficiency. This trend is contributing significantly to the growth of the biologics CDMO secondary packaging market.
Market Challenges
Stringent regulatory requirements
The packaging of biologics must comply with strict regulatory standards, which can be complex and time-consuming. Ensuring compliance across different regions poses a challenge for CDMOs.
High operational and infrastructure costs
Setting up advanced packaging facilities with controlled environments requires significant investment. This can be a barrier for new entrants and smaller players in the market.
Market Segmentation
By Packaging Type
The biologics CDMO secondary packaging market is segmented into cartons, labels, inserts, and others. Cartons hold a significant share due to their widespread use in protecting and organizing biologic products during transportation.
Labels are essential for providing product information and ensuring traceability. Inserts are used to include detailed instructions and regulatory information, enhancing patient safety and compliance.
By Service Type
Based on service type, the market includes labeling, cartoning, serialization, and aggregation. Labeling services are critical for product identification and regulatory compliance.
Cartoning services ensure proper packaging and protection of biologic products. Serialization and aggregation services are gaining importance due to increasing requirements for supply chain transparency and anti-counterfeiting measures.
By End-Use Industry
The market is segmented into pharmaceutical companies, biotechnology firms, and others. Pharmaceutical companies account for a major share due to the high demand for packaging services for biologic drugs.
Biotechnology firms are also significant contributors, driven by the growing development of advanced biologic therapies.
Top Players Analysis
The biologics CDMO secondary packaging market is competitive, with key players focusing on expanding their service offerings and technological capabilities.
- Catalent Inc.
Catalent Inc. is a leading CDMO providing comprehensive packaging solutions for biologics. The company focuses on innovation and global expansion. - Lonza Group AG
Lonza Group AG offers integrated services, including secondary packaging for biologics. The company emphasizes quality and regulatory compliance. - Recipharm AB
Recipharm AB provides contract manufacturing and packaging services, including biologics packaging. The company focuses on flexibility and customer-centric solutions. - PCI Pharma Services
PCI Pharma Services specializes in pharmaceutical packaging and offers advanced secondary packaging solutions for biologics. The company is known for its expertise in serialization and compliance. - Thermo Fisher Scientific Inc.
Thermo Fisher Scientific Inc. provides a wide range of services, including packaging solutions for biologics. The company focuses on innovation and global reach.
Conclusion
The biologics CDMO secondary packaging market is set for significant growth, driven by the increasing demand for biologic therapies and the trend of outsourcing packaging services. The market benefits from technological advancements and the need for compliance with stringent regulations. Despite challenges such as high costs and regulatory complexities, the market offers substantial opportunities for innovation and expansion.