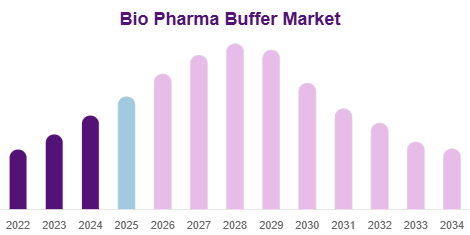
Market Size
The global Bio Pharma Buffer Market size was USD 146.64 billion in 2024 and is projected to grow from USD 155.97 billion in 2025 to USD 238.12 billion by 2033.
The market is expected to grow at a CAGR of 5.43% during the forecast period.
Buy Now : https://reedintelligence.com/market-analysis/bio-pharma-buffer-market/buy-now
Introduction
The Bio Pharma Buffer Market is witnessing significant growth due to the increasing demand for biologics, vaccines, and advanced pharmaceutical manufacturing processes worldwide. Bio pharma buffers are essential chemical solutions used to maintain stable pH conditions during drug formulation, cell culture processing, protein purification, and biopharmaceutical production activities.
According to Reed Intelligence, the growing biotechnology industry, rising pharmaceutical research investments, and increasing production of biologic drugs are major factors driving market expansion. The market is also benefiting from advancements in pharmaceutical manufacturing technologies and rising adoption of single-use bioprocessing systems.
Get Your Sample Report Here: https://reedintelligence.com/market-analysis/bio-pharma-buffer-market/request-sample
Market Dynamics
Market Drivers
Rising Demand for Biologics and Vaccines
The increasing production of biologics, monoclonal antibodies, biosimilars, and vaccines is one of the primary growth drivers of the Bio Pharma Buffer Market. Buffer solutions are widely used in maintaining chemical stability during manufacturing and purification processes.
Growing global healthcare demand and increasing prevalence of chronic diseases are accelerating the need for advanced biologic therapies and pharmaceutical products.
Expansion of Biopharmaceutical Manufacturing
Biopharmaceutical companies are significantly expanding production capacities to meet rising global demand for innovative therapies and vaccines. This expansion is increasing the consumption of pharmaceutical-grade buffer solutions across manufacturing facilities.
The rapid growth of biotechnology research and development activities is further supporting market growth.
Technological Advancements in Bioprocessing
Advancements in automated bioprocessing systems, continuous manufacturing technologies, and single-use systems are improving pharmaceutical manufacturing efficiency. Modern buffer preparation systems provide enhanced consistency, scalability, and operational performance.
These innovations are encouraging pharmaceutical companies to adopt advanced buffer technologies for biologic drug manufacturing.
Increasing Investments in Pharmaceutical Research
Governments, pharmaceutical firms, and biotechnology companies are increasing investments in drug discovery, clinical trials, and healthcare innovation programs. Rising research activities are driving demand for high-quality laboratory and production buffer solutions.
The growing focus on personalized medicine and gene therapies is also contributing to market expansion.
Market Size Link : https://reedintelligence.com/market-analysis/bio-pharma-buffer-market
Market Challenges
High Manufacturing Costs
Bio pharma buffers require highly controlled manufacturing environments and strict quality assurance procedures, which may increase production and operational costs for manufacturers.
Regulatory Compliance Requirements
Pharmaceutical manufacturers must comply with stringent international regulations regarding product quality, sterility, and manufacturing consistency, creating operational challenges.
Complex Storage and Handling Requirements
Certain pharmaceutical buffer solutions require specialized storage conditions and transportation systems, increasing supply chain complexity.
Bio Pharma Buffer Market Segmentation
By Product Type
Phosphate Buffers
Phosphate buffers hold a substantial market share due to their broad application in pharmaceutical formulations and biological research processes.
Acetate Buffers
Acetate buffers are widely utilized in protein purification, biochemical analysis, and vaccine manufacturing applications.
Tris Buffers
Tris buffers are commonly used in molecular biology and biopharmaceutical research because of their effective pH stabilization properties.
Citrate Buffers
Citrate buffers are increasingly adopted in pharmaceutical manufacturing and biologics production applications.
By Application
Drug Formulation
Bio pharma buffers are extensively used in pharmaceutical drug formulation processes to maintain product stability and effectiveness.
Cell Culture Processing
Cell culture applications represent a major segment due to growing biologics production and biotechnology research activities.
Protein Purification
Protein purification processes require advanced buffer solutions for therapeutic protein isolation and biologic compound stabilization.
Vaccine Manufacturing
Buffer systems play a critical role in vaccine production, formulation, and purification operations across pharmaceutical facilities.
By End User
Pharmaceutical Companies
Pharmaceutical manufacturers are major consumers of bio pharma buffers due to increasing biologic drug production and vaccine development activities.
Biotechnology Companies
Biotechnology firms utilize buffer solutions for research, clinical development, and large-scale commercial manufacturing operations.
Research Laboratories
Academic and research institutions use pharmaceutical-grade buffers in molecular biology studies, diagnostics, and drug discovery projects.
Contract Manufacturing Organizations
Contract manufacturing organizations are increasingly adopting advanced buffer systems to support outsourced pharmaceutical production services.
Regional Analysis
North America
North America holds a significant share of the Bio Pharma Buffer Market due to advanced pharmaceutical infrastructure, strong biotechnology research capabilities, and increasing biologics production activities in the United States and Canada.
The presence of leading pharmaceutical companies and rising healthcare investments are supporting regional market growth.
Europe
Europe is witnessing substantial growth driven by increasing pharmaceutical research and expanding vaccine manufacturing operations. Germany, France, and the United Kingdom are key contributors to regional market development.
Asia-Pacific
Asia-Pacific is expected to experience rapid growth during the forecast period due to rising pharmaceutical manufacturing investments, expanding biotechnology sectors, and increasing healthcare infrastructure development across China, India, Japan, and South Korea.
The region’s growing contract manufacturing industry is accelerating market expansion.
Latin America
Latin America is gradually emerging as a promising market due to increasing pharmaceutical production and improving healthcare infrastructure.
Middle East and Africa
The Middle East and Africa region is witnessing moderate growth supported by expanding healthcare investments and growing pharmaceutical manufacturing activities.
Competitive Landscape
The Bio Pharma Buffer Market is highly competitive, with companies focusing on pharmaceutical-grade product quality, advanced manufacturing technologies, and regulatory compliance. Manufacturers are investing in production expansion, research and development, and strategic collaborations to strengthen their market positions.
Companies are also emphasizing customized buffer formulations, automated preparation systems, and scalable production capabilities to meet rising pharmaceutical industry demand.
Top Players Analysis
Thermo Fisher Scientific Inc.
Thermo Fisher Scientific Inc. is a major participant in the Bio Pharma Buffer Market, offering pharmaceutical-grade buffers, laboratory chemicals, and advanced bioprocessing solutions.
Merck KGaA
Merck KGaA specializes in high-purity pharmaceutical buffer systems and advanced manufacturing solutions for biotechnology applications.
Sartorius AG
Sartorius AG focuses on bioprocessing technologies, buffer preparation systems, and pharmaceutical manufacturing equipment.
Lonza Group
Lonza Group provides specialized pharmaceutical manufacturing services and high-quality buffer solutions for biologics production.
Avantor, Inc.
Avantor, Inc. offers laboratory chemicals, pharmaceutical-grade buffer products, and advanced healthcare manufacturing solutions.
Future Outlook
The Bio Pharma Buffer Market is expected to witness continuous growth due to rising biologics production, increasing vaccine demand, and expanding pharmaceutical research activities worldwide. Advancements in bioprocessing technologies and automated manufacturing systems are likely to create substantial market opportunities.
Manufacturers are expected to focus on high-purity formulations, scalable production systems, and innovative buffer preparation technologies to meet evolving pharmaceutical industry requirements. Growing investments in biotechnology infrastructure and healthcare innovation are anticipated to support long-term market expansion.
Conclusion
The Bio Pharma Buffer Market is projected to maintain strong growth momentum due to increasing biologics manufacturing, rising vaccine production, and growing pharmaceutical research investments. Bio pharma buffers continue to play a vital role in ensuring product stability, manufacturing consistency, and operational efficiency within the pharmaceutical and biotechnology industries.
Despite challenges such as regulatory compliance requirements and high manufacturing costs, the market outlook remains positive due to technological advancements and increasing healthcare demand. According to Reed Intelligence, expanding biotechnology applications and rising pharmaceutical innovation are expected to support long-term growth in the global Bio Pharma Buffer Market.
Request Free Sample : https://reedintelligence.com/market-analysis/bio-pharma-buffer-market/request-sample